Alphapointe Secures Patent for Ground-Breaking Splint
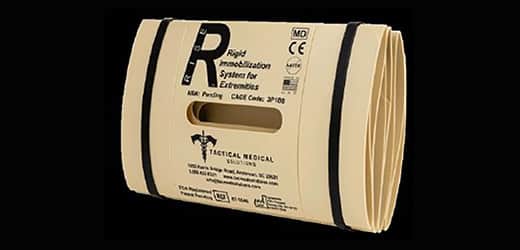
Alphapointe received notice from the United States Patent and Trademark Office that the application for a patent on the RISE Splint has been formally approved.
The patent approval is the latest in a series of developments for Alphapointe’s Tactical/Medical Division, which now has eight patents/patent-pending statuses for devices designed to improve the well-being and safety of people around the world.
“The formal recognition of the patent is the latest step in Alphapointe becoming one of the premier tactical/medical manufacturers in the country,” said Alphapointe President and CEO Reinhard Mabry. “Prior to receiving this status, we had already received orders for tens of thousands of splints, which tells us that we are meeting a critical demand for an innovative product. And, we are thrilled that in creating a unique and essential product that we’re able to provide more great jobs to people who are blind.”
Alphapointe developed the “RISE” (Rigid Immobilization System for Extremities) in conjunction with the U.S. Army. It was tested by military personnel at both Fort Hood, Texas and Schofield Barracks in Oahu, Hawai’i. The splints, which are also used by outdoor enthusiasts who engage in activities such as hiking and mountain climbing, are available to the general public in addition to being used by the military via tacmedsolutions.com/rise.
Earlier this year, Alphapointe entered into agreements with Safeguard Medical and Tactical Medical Solutions to serve as distributors for Alphapointe’s tactical & medical devices. While the tactical & medical products are produced at multiple Alphapointe locations, the splints are manufactured at Alphapointe’s facility in Kansas City with the majority produced by people with vision loss.
“The RISE Splint is one of the most unique tactical/medical devices on the market,” said Alphapointe Chief Operating Officer Ryan Williams. “In signing agreements with Safeguard Medical and Tactical Medical Solutions, Alphapointe has partnered with two of the most prominent tactical/medical distributors in the country.”
The RISE Splint changes the landscape for the application of splints in triage situations, particularly in front line military settings. RISE Splints are significantly more lightweight and compact than predecessors, which allows for non-medical personnel to carry the item and administer critical treatments on the battlefield. The RISE Splint is also unique because it has the capability to be used for both limbs and pelvic stabilization – a feature previously unavailable with standard-issue splints in the U.S. military.
The RISE is the fourth tactical/medical product developed by Alphapointe in the past five years. Previously developed items – largely used by branches of the U.S. military – include the Tactical Mechanical Tourniquet, the Tactical Pneumatic Tourniquet and the Junctional Hemorrhage Control Plate. To date, Alphapointe has produced nearly 500,000 life-saving tactical/medical devices, which has created sustainable jobs for more than 25 Americans, most of whom are people who are blind.
“The initial line of tactical/medical products is just the beginning for Alphapointe,” said Alphapointe Director of the Tactical/Medical Division Christopher Kosiorek, a retired Special Forces Medical Sergeant who served in the military for 23 years. “Alphapointe has 10 additional tactical/medical devices in development, some directly in conjunction with the U.S. Army Medical Research and Development Command. These products will allow us to fill gaps in the American Military and First Responder medical community, with most also available to the general public.”
[embedyt] https://www.youtube.com/watch?v=An5fpTcGb8M[/embedyt]





